

Amphotericin B is the most potent and broad-spectrum antifungal of all the drugs discovered in more than a century of global efforts. Amphotericin B has been in clinical use and has not shown any drug resistance. On one hand owing to its merits it earned the status of a Gold Standard while on the other hand the dose limiting and frequently fatal toxicities predominantly nephrotoxicity labeled it as Ampho- the- Terrible.
Despite the nephrotoxicity in two-thirds of the patients using Amphotericin B, hitherto no better drug could be discovered to replace its broad-spectrum and drug sensitivity- in fact the only antifungal free of drug resistance.
| Product name | Candida | Aspergillus | Mucor | Cryptococcus |
|---|---|---|---|---|
| FUNGISOME® i.v. | Yes | Yes | Yes | Yes |
| Fluconazole | Variable | No | No | Yes |
| Itraconazole | Variable | Yes | No | Yes |
| Voriconazole | Variable | Yes | No | Yes |
| Posaconazole | Yes | Yes | Variable | Yes |
| Caspofungin | Variable | Yes | No | No |
| Anidulafungin | Yes | Yes | No | No |
| Micafungin | Yes | Yes | No | No |
FUNGISOME® i.v. was found to be very stable, safe and highly potent against commonly occurring fungal pathogen. MIC90 of FUNGISOME® i.v. was tested against 262 fungal pathogen clinical isolates. The in vitro activity of FUNGISOME® i.v. appears to be better and has broader-spectrum compared to Conventional Amphotericin B and other Lipid/Liposomal Amphotericin B.
MIC90 of FUNGISOME® i.v. is 2 to 16 times lower than Amphotericin B deoxycholate (Conventional Amphotericin B) for 262 isolates except one Paecilomyces lilacinus. All the isolates of Candida haemulonii are found to be sensitive to FUNGISOME® i.v. with MIC50 of 0.5mg/l, whereas Conventional Amphotericin B is resistant. FUNGISOME® i.v. is found to have broader spectrum of antifungal activity than Conventional Amphotericin B. These findings are in contrast with in vitro studies2,3 conducted with other Amphotericin B formulations, which indicated that other lipid formulations are equally or less active in vitro than Conventional Amphotericin B against Candida spp., Aspergillus flavus, Aspergillus fumigatus and Cryptococcus neoformans.
| Product | Yeast | Filamentous fungi |
|---|---|---|
| Ablecet1 | 0.51 ± 0.21 | 4.34 ± 0.61 |
| AmBisome1 | 1.28 ± 0.24 | 5.68 ± 0.57 |
| Amphotericin B1 | 0.29 ± 0.11 | 1.12 ± 0.19 |
| Product | Yeast | Filamentous fungi |
| FUNGISOME® i.v. 2 | 0.125 (0.03-0.5) | (0.06-2.0) |
| Amphotericin B2 | 0.5 (0.125-16.0) | 0.5-4.0 |
Out of all Amphotericin B and Liposomal Amphotericin B formulations only FUNGISOME® i.v. is effective against
Candida auris and Candida haemulonii
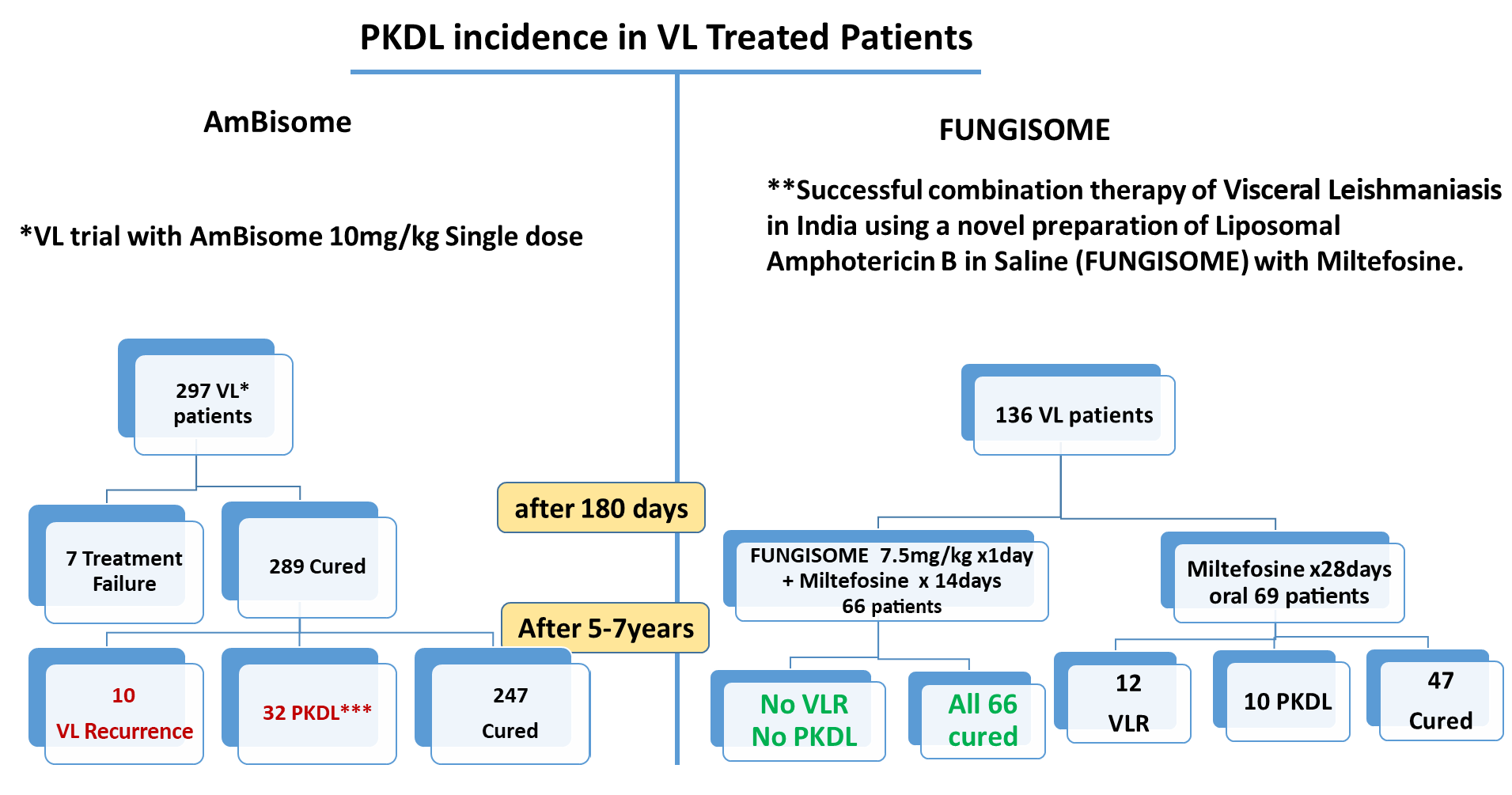
* Mondal et. al. Relationship of Serum Antileishmanial Antibody With Development of Visceral Leishmaniasis, Post-kala-azar Dermal Leishmaniasis and Visceral Leishmaniasis Relapse. Front. Microbiol., 09 October 2019. doi.org
** Goswami R P et al. Combination Therapy against Indian Visceral Leishmaniasis with Liposomal Amphotericin B (FUNGISOME® i.v.) and Short- Course Miltefosine in Comparison to Miltefosine Monotherapy. Am J Trop Med Hyg, doi: 10.4269/ajtmh.19-0931,2020 doi.org
*** Chadha AA et. al. Treatment of Post Kala-Azar Dermal Leishmaniasis with FUNGISOME® i.v. – A Novel Indian Liposomal Amphotericin B. Indian J Drugs Dermatol. 6:28-31,2020. doi.org
| Amphotericin B i.v. formulations | Daily Dose mg (kg/day) | Antifungal Efficacy |
|---|---|---|
| Liposomal Amphotericin B in Saline | 1-3mg/kg/day1,2 |
> 90% Phase II-94.0%3 Phase III- 90.9%3 Phase IV- 91.1%2 |
| Liposomal Amphotericin B (AmBisome & its copies) | 3-6mg/kg/day4,5 | 40-42%7 77 %8 |
| Amphotericin B conventional/ Lipid Emulsion | 1/2 - 5mg/kg/day6 | 31.69 |
| Amphotericin B Lipid Complex | 5mg/kg/day7 | 33.37 |

| Nephro-toxicity | Hepato-toxicity | ||
|---|---|---|---|
| Amphotericin B Formulations | Conventional Amphotericin B | 34%-60%1 | |
| FUNGISOME® i.v./ AmBullet® i.v. |
No Nephrotoxicity reported 2,3,4,5 |
No Hepatotoxicity | |
| Liposomal Amphotericin B | 10%-20% 1 26.9%6 |
||
| Lipid Complex | 42-63% 1 | ||
| Lipid Emulsion | Not reported | Data not available | |
| Echinocandins | Caspofungin | 16.7% 7 | 22.4% 8 |
| Anindulafungin | 37.2% 8 | ||
| Azoles | Micafungin | 23.2% 8 | |
| Fluconazole | 8% 9 | ||
| Itraconazole | 5% 10 | 10% 10 | |
| Voriconazole | 7% 11 | 19.2% 12 | |
| Posaconazole | 5%-11% 13 |
FUNGISOME® i.v. - A Patented Innovative Liposomal Amphotericin B in Saline Most Nephro-safe and Effective Formulation